
Close

Retatrutide represents the latest frontier in metabolic and endocrine research. It belongs to a sophisticated new class of multi-receptor peptide agonists designed to interact with several metabolic signaling pathways simultaneously.
While earlier therapeutic strategies focused on activating a single receptor—specifically the [GLP-1 (Glucagon-Like Peptide-1)] pathway—recent scientific work explores peptides that activate multiple receptors to investigate broader, synergistic metabolic responses. Retatrutide is currently one of the most widely studied compounds in this category of «triple-agonist» metabolic peptides.
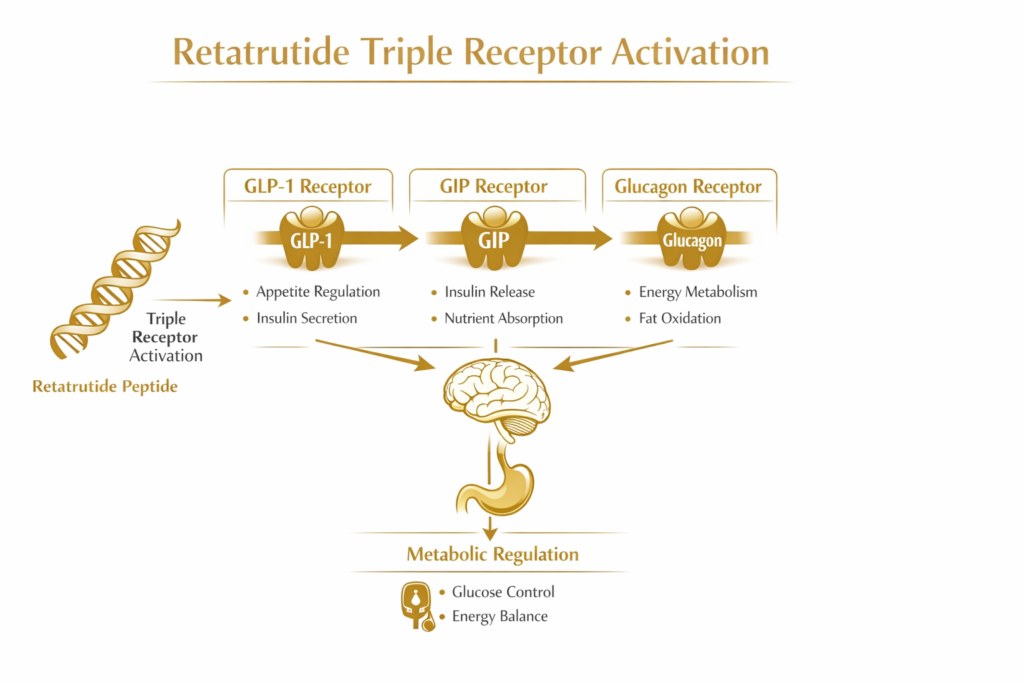
Retatrutide is a synthetic peptide engineered to activate three distinct metabolic receptors involved in incretin signaling:
GLP-1 Receptor: Associated with appetite regulation and glucose-dependent insulin signaling.
GIP Receptor: Associated with nutrient sensing and incretin hormone activity.
Glucagon Receptor: Associated with energy metabolism and hepatic (liver) metabolic signaling.
Because the molecule interacts with these three pathways simultaneously, it is described in scientific literature as a triple-receptor incretin agonist. This multi-pathway approach allows researchers to observe how simultaneous activation influences metabolic regulation differently than compounds targeting a single or dual pathway, such as [Tirzepatide].
Retatrutide works through receptor-mediated signaling rather than hormone replacement. By binding to these three receptors, it influences the complex communication between the gut, pancreas, liver, adipose tissue, and the central nervous system.
This «Triple Signaling» model is a prime example of the precision modulation discussed in [Peptides vs. Hormones: Biological Signaling]. Rather than overriding the endocrine system, Retatrutide fine-tunes the body’s existing nutrient-sensing machinery to observe changes in energy expenditure and glucose disposal.
One of the most significant findings in recent clinical sub-studies (published in Nature Medicine) is Retatrutide’s impact on MASLD (Metabolic Dysfunction-Associated Steatotic Liver Disease). Research indicates that over 85% of subjects on higher doses (8mg–12mg) reduced their liver fat to under 5%, effectively «clearing» the diagnostic criteria for fatty liver within 48 weeks.
In leading clinical studies, such as the TRIUMPH-1 trials published in the New England Journal of Medicine, the Retatrutide dosage follows a specific monthly «titration» or escalation schedule. This gradual approach is designed to allow the body’s triple-receptors (GLP-1, GIP, and Glucagon) to adapt comfortably while minimizing gastrointestinal sensitivity.
Month 1 (Initiation): Typically begins at 2mg per week to establish a metabolic baseline.
Month 2 (Escalation): Increased to 4mg per week as appetite regulation stabilizes.
Month 3 & Beyond (Maintenance): Depending on research goals, doses may scale to 8mg or 12mg per week for maximum metabolic effect.
Important Note: At Myosfit, we emphasize that any protocol involving Retatrutide should be monitored by a healthcare professional. To protect lean muscle mass during rapid weight regulation, we often recommend pairing Retatrutide with supportive peptides like BPC-157.
When searching for where to buy Retatrutide in Mexico, it is vital to distinguish between «gray-market» resellers and verified laboratory environments. As a COFEPRIS-registered laboratory, Myosfit provides pharmaceutical-grade compounds that prioritize purity and safety.
The price of Retatrutide reflects the rigorous standards of a professional laboratory. While «research chemicals» may vary in consistency, Myosfit protocols (such as our Hollywood Stack) are priced to include:
99%+ Verified Purity: Ensuring zero fillers or contaminants.
Synergistic Support: Our stacks include GHK-Cu or BPC-157 to ensure your skin and joints stay resilient during your transformation.
Secure Local Logistics: Fast overnight shipping available within Mexico to maintain peptide integrity.
In the context of advanced research, Retatrutide’s ability to influence energy regulation is often studied alongside a subject’s foundational health.
One of the primary areas of investigation is how triple-agonists affect body composition. Because of the intense metabolic shift and rapid weight regulation observed in Retatrutide research, scientists often evaluate the need for muscle-preserving strategies. As explored in [Longevity and Strength: Why Muscle Matters for Aging], maintaining lean tissue is critical during periods of rapid fat loss.
Furthermore, research models often investigate whether a functional hormonal environment, such as the one discussed in [Menopause and Hormone Health], enhances the metabolic efficiency of multi-receptor agonists like Retatrutide.
Simplified signaling model
Retatrutide peptide activates GLP 1 receptor
Retatrutide peptide activates GIP receptor
Retatrutide peptide activates glucagon receptor
Activation of these receptors influences metabolic communication between the gut, pancreas, liver, adipose tissue, and areas of the central nervous system involved in appetite and energy regulation.
Molecular Engineering
The structure of retatrutide includes amino acid substitutions and lipid side chain modifications designed to extend circulating half life and enhance receptor interaction.
These modifications are commonly used in peptide pharmacology to improve stability and reduce rapid enzymatic degradation that occurs with naturally occurring incretin hormones. By modifying the peptide backbone and attaching fatty acid groups, researchers create molecules that maintain receptor signaling activity for longer periods.
A randomized controlled trial investigated the biological effects of retatrutide in adults with obesity or overweight.
Participants were assigned to different dose groups and observed over several months while researchers evaluated changes in metabolic markers and body weight. The study examined dose response relationships, safety observations, and changes in metabolic parameters associated with incretin signaling pathways.https://pmc.ncbi.nlm.nih.gov/articles/PMC12767911/
Following earlier studies retatrutide entered large scale global research programs designed to evaluate longer observation periods and broader populations.
Phase 3 studies typically investigate outcomes in thousands of participants across multiple research centers. https://investor.lilly.com/news-releases/news-release-details/lillys-triple-agonist-retatrutide-delivered-weight-loss-average
These trials evaluate long term metabolic outcomes, safety monitoring, and durability of biological effects.
Clinical research programs include investigations involving metabolic disorders related to obesity, long term weight regulation pathways, metabolic dysfunction associated with fatty liver disease, and endocrine signaling related to glucose metabolism.
Future Directions of Multi Receptor Peptide Research
The development of triple receptor agonists represents a major direction in metabolic peptide research.
Scientists continue exploring whether activating several incretin pathways simultaneously may influence energy regulation differently than compounds targeting a single receptor.
Ongoing investigations are evaluating multi agonist peptides in metabolic medicine, endocrinology, and obesity related research in order to better understand complex hormonal signaling networks involved in metabolism.
Regulatory Context in Mexico
In Mexico peptide based compounds may fall under different regulatory classifications depending on formulation and distribution framework. Oversight of pharmaceutical and biologic products is managed by COFEPRIS.
Professional peptide distribution emphasizes traceable sourcing, laboratory documentation, product quality verification, and regulatory compliance aligned with national regulatory standards.
Scientific References
New England Journal of Medicine Retatrutide Study https://www.nejm.org/doi/full/10.1056/NEJMoa2301972
PubMed Clinical Trial Summary https://pubmed.ncbi.nlm.nih.gov/37366315/
Clinical Trials Registry https://clinicaltrials.gov/search?cond=&term;=retatrutide

Professionally sourced peptides with an emphasis on quality, documentation, and responsible handling.

• What peptides really are and how they function inside the body
• The logic and structure behind high-performance MyosFit stacks
• Fat loss, muscle recovery, sleep, longevity, and skin optimization strategies
• How advanced stacks are engineered, not guessed
• What defines clinical-grade peptides,s and why most products fail that standard
The date has not been announced yet – join the early interest list to be notified first when registration opens.
Signup for news and special offers!
You have successfully joined our subscriber list.